
Figure 1: Quantum dot technology is used for diagnostic techniques
Improvements in manufacturing precision have resulted in smaller, more efficient sensors. Integrating these advances with communication technologies, specialists or automated systems can interpret diagnostic data remotely, bridging geographical and economic divides.
Light-based technologies
Progress in light-based technologies, including lasers, LEDs and photodiodes, has been critical in transforming diagnostics. GaN was introduced initially for bluer wavelengths and then, through phosphor excitation, for yellow wavelengths. Due to its wide band gap, it is now used for a broad range of wavelengths at high efficiencies.
The development of GaN lasers was driven by commercialisation of the Blu-ray player, which drove efficiency and cost reduction at 405nm for several years. The development of fluorescent dyes for protein tagging excited at this wavelength is now widely used for fluorescence microscopy, gene sequencing and anemometry, for example.
In the past decade quantum dot technology has moved from research labs to industrial adoption for LED enhancement. Quantum dots are nanoparticles, ranging from ~2-10nm in diameter, which emit light at specific wavelengths determined by their size. This effectively gives a high efficiency emitter at a very well controlled wavelength. Historically, fluorescent dyes were developed based on available wavelengths of LEDs/lasers. Now LEDs, diode and diode-pumped solid state lasers are being developed to match the excitation wavelengths of the dyes, for gene sequencing, cytometry and other diagnostic techniques (see Figure 1).
Advances in cameras allow data to be captured at ever-increasing rates. In one form of gene sequencing, vast samples of fragments of DNA are imaged using fluorescent tags at four wavelengths and repeated patterns of nucleotides are identified as being from the same location on the genome. As the patterns overlap a model of the complete genome is put together.
For the camera to collect the same data at a higher frame rate and over a wider field of view, the fluorescence imaged needs to be brighter, which calls for more optical power. The cost of fluorescent dye drives demand for high-speed, high-resolution cameras to work at lower light levels.
Along with LEDs and lasers, photodiodes and charge-coupled devices are becoming more efficient through innovations in materials and design. Enhanced materials improve quantum efficiency. The use of GaN has significantly advanced detection at optical wavelengths. Optimisation of semiconductor doping, including gradient doping across the semiconductor, surface doping through atomic layer deposition and surface passivation (essentially coating the semiconductor surface to reduce defects), has allowed further gains in quantum efficiency, enabling improved performance in low-light applications.
Clinical applications
Photonics has also transformed clinical tools. Once limited to research institutions, femtosecond and supercontinuum lasers are now used in optical coherence tomography for routine eye health checks (Figure 2). This progress is made possible by the physics of lasers, where the coherence length of a laser is inversely proportional to its bandwidth. Broad pulse bandwidths allow measurements equal to retina thickness. Using a tuneable wavelength femtosecond laser or a filtered supercontinuum source, opticians can create 3D macula maps for early detection of conditions such as macular degeneration.
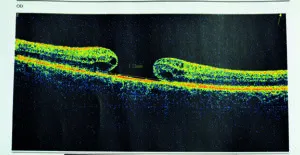
Figure 2: Image of the eye created using OCT
Real-time data and the role of AI
Advances in sensor technology have accelerated processes such as DNA sequencing. The first human genome was sequenced at a cost of approximately $2.7bn; today, personal DNA sequencing can be completed in minutes for tens of dollars.
High throughput systems allow more samples to be analysed and reduce cost. In flow cytometry, where cells are imaged and sorted in real time, applications are broadening from diagnostics to (for example) sperm cell sorting to produce a higher viability sample by either positive selection, or ablation of non-viable cells.
AI manages the ever-increasing data gathered from wearable diagnostic sensors. It allows personalisation through interpreting data in the context of an individual’s medical history, including genetic information and with improvements in accuracy through adaptive learning (subject to privacy restrictions). It is easy to envisage a transition from patient monitoring to drug specification based on this data, with continually optimised dosages rather than a course of standard ones. This offers improved patient outcomes and cost savings on expensive drugs. A further development is likely to be automated drug administration, to control chronic diseases such as diabetes, and for high-risk patients with heart disease or severe allergies.
Overcoming challenges
Despite remarkable advances, sensor technology faces significant challenges. The greatest is environmental variability. For the interpretation of fluorescence and other imaging techniques to be effective, the relative intensity needs to be understood with varying sample size, health, temperature and concentration, before effects on the sensor due to temperature, humidity, loss of efficiency or other calibration drift are accounted for.
EMI, vibration and other noise sources as well as human movement of changes, can give instantaneous erroneous results, necessitating advanced filtering and error-correction algorithms. In many cases this can be resolved, or at least improved, through the use of further sensors to control or account for the changing environment and performance of the primary sensor.
Data management also presents significant challenges. Continuous monitoring generates massive volumes of data that require efficient storage, secure transmission and timely analysis.
The cost of implementing advanced sensors remains a barrier in many regions. While the affordability of diagnostic equipment has improved, the initial investment can be prohibitive for small clinics or low-income regions.
Putting the responsibility for when and how diagnostic instruments are used onto the patient requires the patient to be sufficiently willing, capable and organised to operate the instrument correctly. If the instrument presents data to the patient, it must be explained and given context to ensure that it does not create upset, misinterpretation or an unwanted change in behaviour. It may also make the patient feel over-confident and reticent to keep in regular contact with their healthcare provider.
The future of sensors
Continual reduction in size and cost of sensors is paving the way for increased wearable and implantable diagnostic tools. Implantable or wearable glucose monitors for diabetes care, for example, will allow better condition monitoring and changes to dosage and timing of insulin administration and may ultimately lead to continuous administration with intelligent dosage control. The same could apply to the rate of administration of chemotherapy (and other treatments) based on variations of the patient’s vital signs and the concentration of cancer markers in the bloodstream or other real-time diagnostics. Another key aspect remains the real-time interpretation of this data. As the early stages of commercialisation of quantum computers come to fruition, the scope for AI-led advances in diagnostics is huge.
 Electronics Weekly
Electronics Weekly



